
Dr. Sameera R. Gunatilake
Grad. Chem., Ph.D. (Mississippi State),
C.Chem., M.I.Chem. C.
Welcome to Gunatilake Group!
“Discovery is the endless exploration of knowledge.”
Welcome to the research group of Dr. Sameera R. Gunatilake at the College of Chemical Sciences, Institute of Chemistry Ceylon.
Our research focuses on the development of engineered biomass-derived carbonaceous materials for environmental remediation and sustainable agricultural applications. A major component of this work involves understanding the physical chemistry of adsorption, including adsorption mechanisms, kinetics, thermodynamics, and material regeneration. We are particularly interested in designing nanoscale iron–biochar composite systems for enhanced contaminant removal and investigating the mechanistic interactions governing their performance.
In parallel, our group works on developing novel analytical methods for the determination of trace-level organic and inorganic analytes in environmental and food matrices, as well as studying the occurrence, transport, and fate of trace contaminants in natural systems.
Our research also extends to chemical education, where we focus on developing innovative pedagogical approaches, exploring theoretical frameworks in chemistry learning, and translating educational philosophy into effective classroom practice and assessment strategies.
Our research group actively welcomes impactful interdisciplinary collaborations. We also encourage motivated and innovative students and researchers to connect with us and explore opportunities to contribute to our work. For further information, please feel free to contact us at ranmal@ichemc.edu.lk.

About Sameera
I am an academic and a professional chemist with an analytical chemistry background by training. I am an expert in chromatographic and spectroscopic analytical method development and quality assurance for trace level pollutants in environmental matrices. Furthermore, in the field of materials chemistry, I have contributed to the area of engineered biochar materials and their iron based nano composites for environmental remediation and agricultural soil amendment. Including material synthesis, characterization and applications.
I have over a decade of experience as a senior academic in tertiary education, involving university lecturing, curriculum development, student administration and research supervising.
Teaching
Dr. Gunatilake is currently on sabbatical leave and therefore does not have active teaching assignments at the Institute of Chemistry Ceylon.
His areas of teaching expertise include:
- Level 1 (Freshman): Titrimetry, Atomic Structure, and Quantum Theory
- Level 2 (Sophomore): Separation Methods, Analytical Atomic Spectrometry, Basic Quantum Mechanics, and Environmental Chemistry
- Level 3 (Junior): Advanced Instrumental Analysis and Advanced Chromatography
- Level 4 (Senior): Green Chemistry, Environmental Modeling, and Chemical Education
In addition, he conducts laboratory classes in Inorganic and Analytical Chemistry.
Research
Biochar
Biochar is a porous carbon-rich material produced through the thermal decomposition of biomass under oxygen-limited conditions, a process known as pyrolysis. Owing to its highly developed pore structure and diverse surface functional groups, biochar exhibits strong adsorption capabilities toward a wide range of organic and inorganic contaminants.
These adsorption properties arise from multiple interaction mechanisms, including pore filling, electrostatic attraction, hydrogen bonding, surface complexation, and π–π electron donor–acceptor interactions. The efficiency of contaminant removal depends strongly on the physicochemical characteristics of biochar, such as surface area, pore size distribution, surface charge at the operational pH, polarity, and the abundance and type of functional groups.
Beyond water remediation, biochar has gained significant attention as a soil amendment due to its ability to enhance crop productivity and improve soil health. It can contribute nutrients directly, improve fertilizer retention and slow nutrient release, and positively influence soil properties such as pH, bulk density, carbon-to-nitrogen ratio, and water holding capacity. In addition, biochar can help reduce the uptake of toxic heavy metals by plants.
The physicochemical properties of biochar are primarily governed by feedstock selection, production conditions, and post-synthesis modifications. These tunable characteristics make biochar a versatile platform material for environmental applications, including serving as a stabilizing support for engineered nanoparticles by reducing their aggregation and surface passivation.
Biochar has emerged as an important research focus due to its low production cost, utilization of abundant biomass and waste resources, relative simplicity of preparation, contribution to sustainable waste management, and potential role in long-term carbon sequestration.
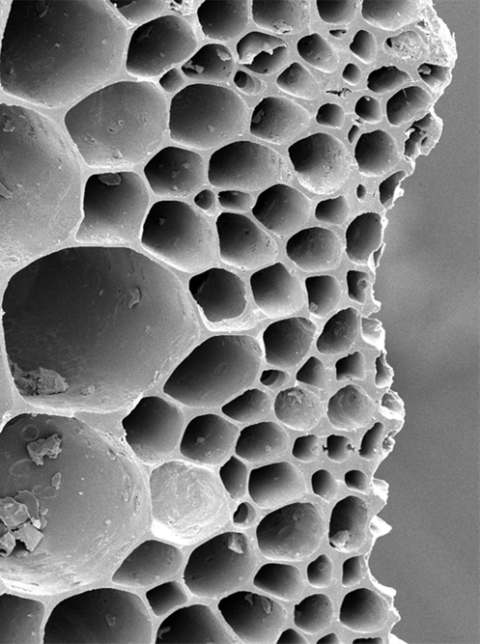
SEM image of tea-waste biochar showing a honeycomb-like porous morphology.
Engineering biochar for enhanced efficiency
Biochar performance can be significantly enhanced through targeted engineering strategies that modify its surface chemistry and pore structure. Such modifications may be carried out either before or after pyrolysis, with post-pyrolysis treatments being more commonly employed to tailor adsorption properties for specific contaminants.
Engineering approaches include both chemical and physical treatments. Chemical modification methods such as mineral acid, alkali, and oxidizing treatments can alter surface functional groups, remove inorganic mineral phases, and modify pore accessibility. Physical treatments including steam activation and plasma processing can further enhance porosity and surface reactivity.
Mineral acid modification is considered one of the simplest and most cost-effective chemical engineering approaches. Acid treatments can substantially influence the structural and surface characteristics of biochar by promoting demineralization, increasing pore volume, and enhancing surface heterogeneity. These changes often improve adsorption efficiency by facilitating pore filling and strengthening adsorbate–surface interactions.
Our research focuses on systematically investigating how mineral acid modifications influence biochar physicochemical properties and adsorption performance. By understanding these relationships, we aim to develop designer biochars tailored for efficient and selective water remediation applications.
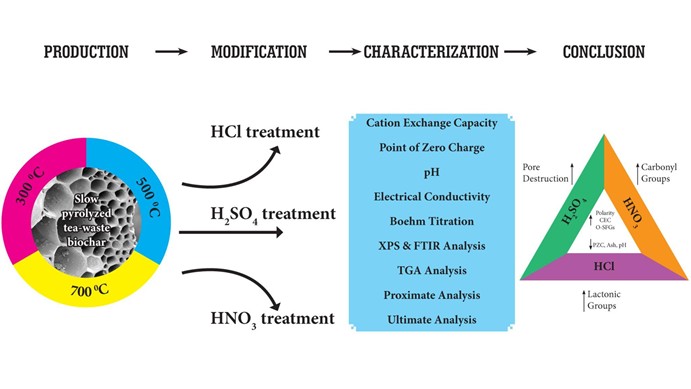
Effects of mineral acid modification on biochar: experimental design and conclusions
Nanoscale Magnetite–Biochar Composites
Nanoscale magnetite–biochar composites represent a promising class of functional materials for environmental remediation in aqueous systems due to their combined adsorption capacity, redox activity, and magnetic separability. Incorporating iron oxide nanoparticles into porous biochar matrices enables the development of engineered adsorbents with enhanced surface reactivity and improved material recovery following treatment.
Our studies show that magnetite incorporation can significantly enhance remediation performance by providing additional reactive binding sites and promoting surface-mediated redox transformations of certain contaminants. These combined adsorption and transformation processes contribute to improved removal efficiency for a range of environmentally relevant pollutants.
Our research focuses on understanding how magnetite incorporation influences adsorption mechanisms and removal efficiency toward both organic and inorganic contaminants. Through these efforts, we aim to develop tunable composite systems with improved selectivity, performance, and operational feasibility for sustainable water remediation applications.
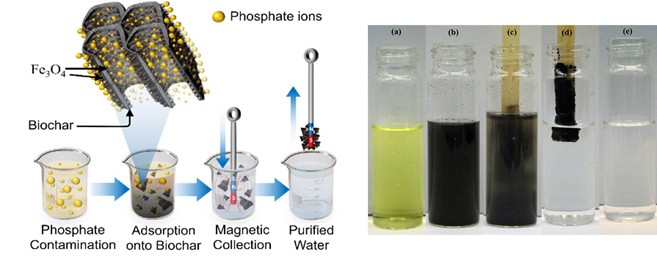
Encapsulation engineering of nZVI–carbon composites for aqueous contaminant remediation
Nanoscale zero-valent iron (nZVI) is widely explored for environmental remediation due to its strong reducing capability, high surface reactivity, and relatively low cost. However, pristine nZVI particles are prone to rapid surface passivation and aggregation, which can limit their stability and performance in aqueous systems. Biochar has therefore gained attention as an effective stabilizing support, where its porous structure and surface functionality improve nanoparticle dispersion, reduce agglomeration, and enhance contaminant accessibility. As a result, nZVI–biochar composites (nZVI@biochar) have emerged as promising materials for developing efficient and sustainable remediation technologies.
Our findings indicate that nZVI@biochar composites synthesized via chemical and carbothermal reduction routes exhibit distinct nanostructural characteristics and stability behaviour. In particular, the composition and nature of the core–shell encapsulation surrounding nZVI play a critical role in determining material performance. We are interested in further investigating remediation mechanisms and understanding how shell structure influences stability and reactivity toward both inorganic and organic pollutants.

HR-TEM images showing the core–shell structure of nZVI with graphene encapsulation
